If you manage gloves for a clinic, a cold‑chain food facility, or a jan‑san distribution network, you’ve probably felt the squeeze: prices swing, freight fluctuates, and consumption spikes when policies drift. The fastest way to reduce spend isn’t chasing the lowest unit price—it’s building a procurement program that keeps quality intact while pulling proven cost levers you can control. This guide walks through those levers, shows how U.S. standards map to purchasing decisions, and closes with a practical, neutral product example for high‑volume environments.
Think in Total Cost of Ownership, Not Unit Price
Price per box is easy to quote, but it rarely tells the whole story. Total Cost of Ownership (TCO) includes the costs you incur because gloves fail early, are changed too often, ship inefficiently, or don’t match the task. That’s why two gloves at the same unit price can deliver very different “cost per use.”
Key drivers include changeout frequency by task, defect‑related waste (holes or tears out of the box), pack‑count and cartonization, freight and lead times, and whether the material is over‑ or under‑spec’d for the job. Tightening any one driver helps; optimizing all of them compounds savings without compromising safety.
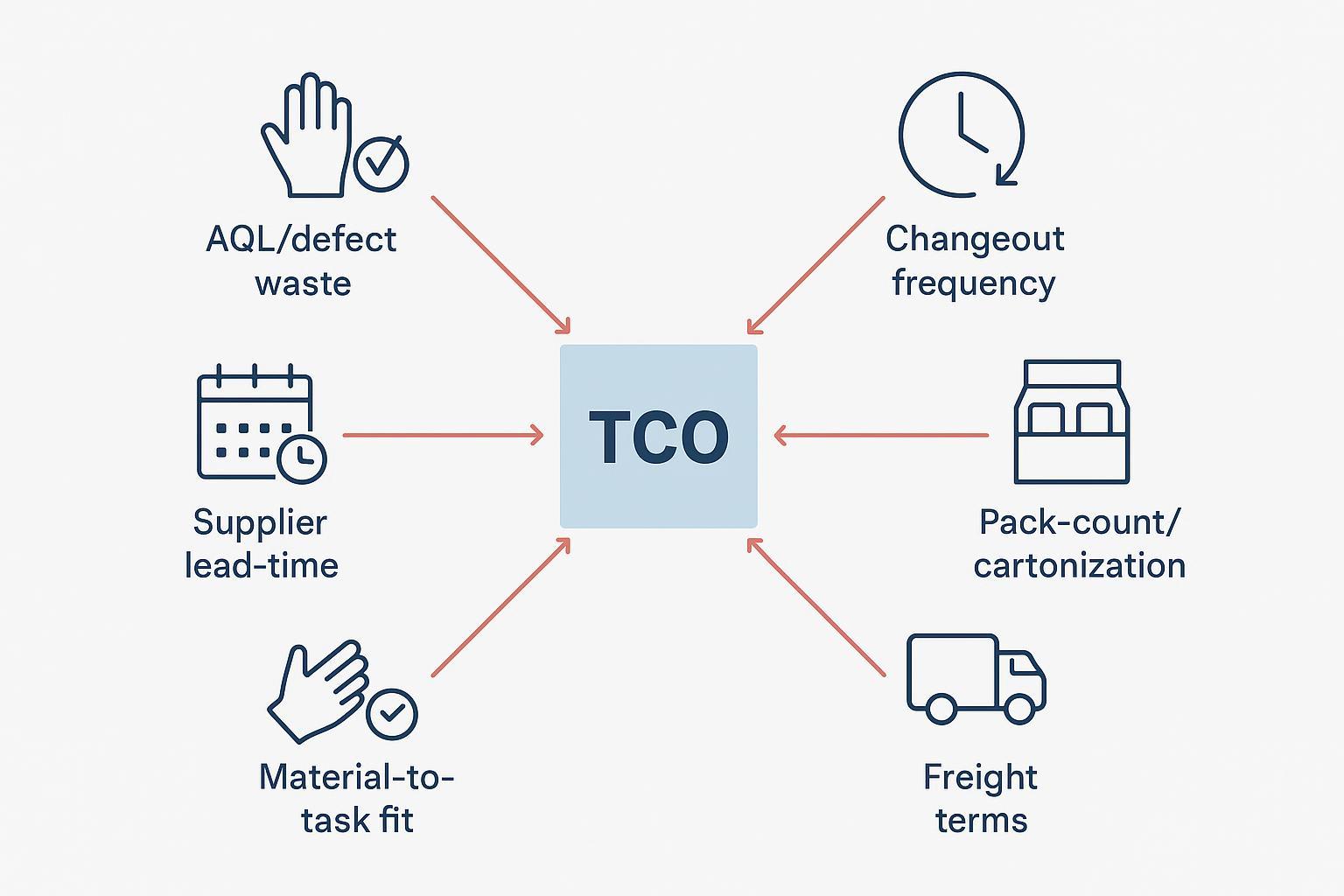
Think of TCO as a set of dials. If your AQL and incoming quality controls reduce defect waste by half, and your policy update trims unnecessary mid‑task changes, the savings stack. Meanwhile, cube‑efficient packaging and stable lead times cut soft costs like expedites and stockouts.
Know the Categories and the U.S. Compliance Basics
Glove materials behave differently under stress, solvents, and temperature, and those differences show up in cost and performance. Matching material to task—within the boundaries of U.S. regulations—keeps quality and safety intact while you trim spend.
Materials at a glance and fit‑for‑task
-
Nitrile examination gloves generally provide strong puncture resistance, broad chemical tolerance, and high elasticity—appropriate for clinical use and higher‑risk industrial tasks. They often cost more but can reduce changeouts where durability matters.
-
Vinyl (PVC) examination gloves typically cost less, with lower elasticity. They can be fit‑for‑purpose in lower‑risk, short‑duration tasks where dexterity and prolonged tactile sensitivity are less critical. Modified‑vinyl formulations can improve stretch and comfort while remaining cost‑efficient.
-
Latex and other elastomers (e.g., polychloroprene, polyisoprene) have specific performance profiles but may be limited by allergy policies and price. Many U.S. healthcare buyers favor nitrile for this reason.
Whichever path you choose, ensure the glove’s labeling and documentation match the application (exam‑grade vs. food handling vs. utility).
What 510(k) and ASTM really mean for purchasing
For patient examination use in the U.S., medical gloves are regulated devices. Procurement should verify exam‑grade status through 510(k) documentation and ensure the product conforms to recognized standards referenced in those submissions.
-
The FDA’s public 510(k) database lets buyers verify exam‑glove clearances and claims; it’s a practical cross‑check during sourcing. See the searchable resource in the FDA’s premarket notification database for exam gloves and the agency’s recognized standards entry for ASTM D5151 water‑leak testing, which underpins “freedom from holes.”
-
OSHA requires appropriate gloves whenever contact with blood or other potentially infectious materials is anticipated; gloves must be replaced when torn or contaminated and never washed or reused. See OSHA’s Bloodborne Pathogens Standard, 29 CFR 1910.1030.
-
During shortages, CDC/NIOSH documented conservation practices that can inform right‑sized policies without compromising safety. Review NIOSH’s guidance on conserving disposable gloves so your policies align with evidence‑based approaches.
-
When scanning specs, you’ll often see ASTM identifiers: D6319 (nitrile), D5250 (vinyl), D5151 (water‑leak test), and, when applicable, D6978 (chemotherapy drug permeation) or F1671 (viral penetration). These tell you which performance and test criteria the product targets.
Practical Levers to Reduce Glove Procurement Costs Safely
Usage audits and right‑sized changeouts
Map high‑consumption tasks by department and shift. Compare observed changeout habits with OSHA indications and your infection control policies. In clinics and labs, bundle sequences of compatible tasks to avoid unnecessary mid‑procedure changes, while keeping hand hygiene intact. In food processing, align glove changes to HACCP checkpoints. Referencing CDC/NIOSH conservation concepts helps teams update SOPs confidently.
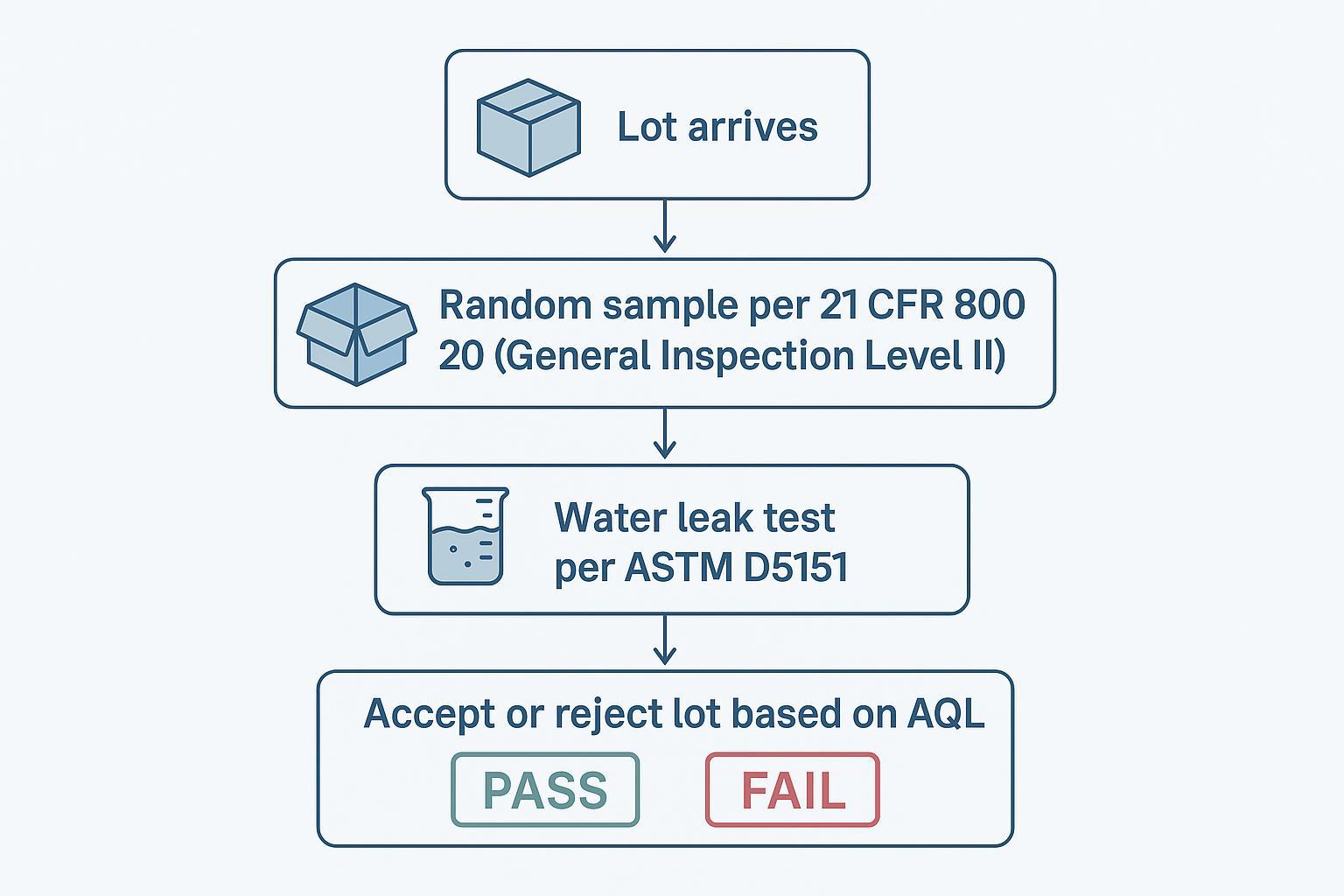
AQL, defect rates, and waste reduction
AQL (Acceptable Quality Level) captures the maximum percentage of defective items (e.g., pinholes) expected in a lot. For exam gloves, FDA sampling plans in 21 CFR 800.20 use the ASTM D5151 water‑leak test. If you’re seeing frequent out‑of‑box tears or leaks, you’re paying for waste and rework. Tighten your spec to reflect an AQL appropriate to your risk posture (2.5 is typical for exam gloves) and require vendors to provide quarterly lot performance and any import‑alert notices. Small improvements in incoming quality can unlock outsized savings in high‑volume settings.
Pack‑count and freight: cartons, cube, and UoM alignment
Standardize your unit of measure (each/box/case/pallet) across ERP and supplier catalogs, then align orders to full‑case or pallet multiples to improve cube utilization and reduce split‑case handling. Confirm GTINs at every packaging level to cut mispicks and returns. In distribution channels, carton density targets and stable case counts reduce both freight variability and receiving labor.
Contracting levers: price tiers, MOQs, lead time, private label
Use volume‑tiered pricing with commitment windows, and write lead‑time SLAs with performance guardrails. For exam‑grade items that carry special claims (e.g., chemotherapy drug resistance), require evidence aligned to the applicable ASTM practice. If private label matters to you, confirm regulatory responsibilities up front (510(k) ownership/listing and labeling compliance) and evaluate whether the manufacturer’s capacity can support your forecast without service erosion.
Worked Examples: Cost per Exam Glove vs. Cost per Use
Let’s put numbers to it. Imagine a community clinic buying 2 million exam gloves annually at $0.055 per piece. Incoming inspections show 2.5% defect‑related waste (pinholes or tears), and observations reveal 10% of changes are mid‑task and avoidable with a policy tweak.
-
Baseline annual cost: $110,000 for 2,000,000 pieces. Waste writes off 50,000 gloves up front; avoidable changes account for another 200,000. Effective useful uses: 1,750,000, so cost per use is $110,000 / 1,750,000 ≈ $0.0629.
-
After interventions: Tighten vendor QA (same price) to reduce true waste to 1.5% and update SOPs to cut avoidable changes to 5%. Effective useful uses rise to about 1,890,000, pushing cost per use to ~$0.0582—roughly a 7.5% improvement without touching the unit price.
A cold‑chain food plant sees different dynamics. At −18°C, low elasticity can cause more tear‑on‑donning events. Suppose baseline waste from low‑temp brittleness is 4% on a $0.040 glove. A trial with a more elastic, exam‑grade modified‑vinyl reduces low‑temp tear events to 2%. On 10 million gloves, that’s 200,000 extra successful uses—$8,000 in avoided waste—plus soft savings from fewer change pauses and better line continuity. The point isn’t that one material is always cheaper; it’s that the right material‑to‑task fit reduces changeouts and defects, which lowers cost per use.
Supplier Vetting and the Scorecard You’ll Reuse
Use a simple, repeatable rubric to avoid surprises at scale:
-
Verify exam‑grade status in the FDA 510(k) database and align claimed standards (e.g., D6319 or D5250; D5151; D6978 if labeled for chemo).
-
Request historical lot‑level defect data and any import alerts; require quarterly summaries.
-
Confirm QMS credentials (e.g., ISO 13485), packaging GTINs, and consistent pack counts across SKUs.
-
Trial gloves in the highest‑consumption tasks and environments first (e.g., cold rooms, sanitation shifts) and track tear‑on‑donning and mid‑task change rates.
-
Lock in lead‑time expectations with measurement and remedies; align cartons to your storage and freight cube.
If you need a large‑scale manufacturing partner with global capacity and private‑label support, shortlist established producers. For example, see how a mature disposable gloves manufacturer presents product categories, packaging, and documentation to assess fit at a glance.
Where a Modified‑Vinyl Exam Glove Can Help
In high‑volume, lower‑risk tasks that still require exam‑grade documentation—think routine examinations, food prep stations away from harsh solvents, or jan‑san wipe‑down workflows—an exam‑grade modified‑vinyl can lower unit cost while protecting usability metrics like fit and stretch. One example is INTCO’s Synmax Pro exam gloves. According to manufacturer‑reported data, Synmax Pro uses a chemically modified vinyl‑based formulation designed for a nitrile‑like fit, with 400%+ verified elasticity in internal testing and elevated abrasion performance compared with conventional nitrile references. They’re powder‑free, non‑sterile, and latex‑free, available in S–XL and multiple colors.
Procurement note: When you evaluate any modified‑vinyl for exam use, confirm that labeling, 510(k) documentation, and referenced ASTM methods match your risk context (e.g., D5250 for vinyl‑family exam gloves and D5151 for leak testing). In cold‑work or food logistics, add a short operational trial at target temperatures to validate tear‑on‑donning rates and grip performance before committing tiers.
Next Steps
Reducing glove spend without compromising quality is about disciplined procurement: verify exam‑grade claims in authoritative databases, target the right AQL and incoming QA, align packaging to your logistics, and choose materials that fit the job. If you’d like background on a global producer’s footprint and quality systems, review INTCO Medical’s company overview and the INTCO Medical corporate site. For RFPs, private‑label inquiries, or documentation requests, you can also contact the manufacturer to discuss specifications, pack counts, and trial protocols that support cost‑per‑use improvements.
FAQ
Q1: What’s the difference between “medical grade” and “FDA 510(k) cleared” for exam gloves?
A: In U.S. healthcare contexts, patient examination gloves are medical devices; exam‑grade products should be listed with the FDA and typically have a 510(k) clearance demonstrating substantial equivalence, including performance and biocompatibility testing. Always verify in the FDA’s public database.
Q2: What does AQL 2.5 mean in practical terms?
A: It’s the maximum defect percentage a sampling plan is designed to accept for a lot (for exam gloves, pinholes detected by water‑leak testing). Lower AQL targets can reduce out‑of‑box waste but may carry pricing or capacity trade‑offs; choose based on risk and trial data.
Q3: When is vinyl acceptable in food handling?
A: Many food‑service tasks can use vinyl or modified‑vinyl gloves if they meet relevant food‑contact requirements and deliver adequate dexterity and tear resistance for the station. For cold rooms or tasks with higher abrasion, trial an exam‑grade, more elastic option to reduce tear‑on‑donning events.
Q4: What’s required to private‑label exam gloves in the U.S.?
A: Clarify who holds the 510(k) and who is responsible for labeling compliance and listing. Ensure QMS coverage (e.g., ISO 13485), consistent packaging identifiers (GTINs), and service levels (lead time, fill rate) before you switch.
Q5: How do pack counts affect cost?
A: Inconsistent inner‑pack or case counts drive mispicks, partials, and freight inefficiency. Standardizing counts and aligning orders to case/pallet multiples improves cube utilization and reduces touches—lowering indirect costs.







